Clinical Trial Supplies Market Overview
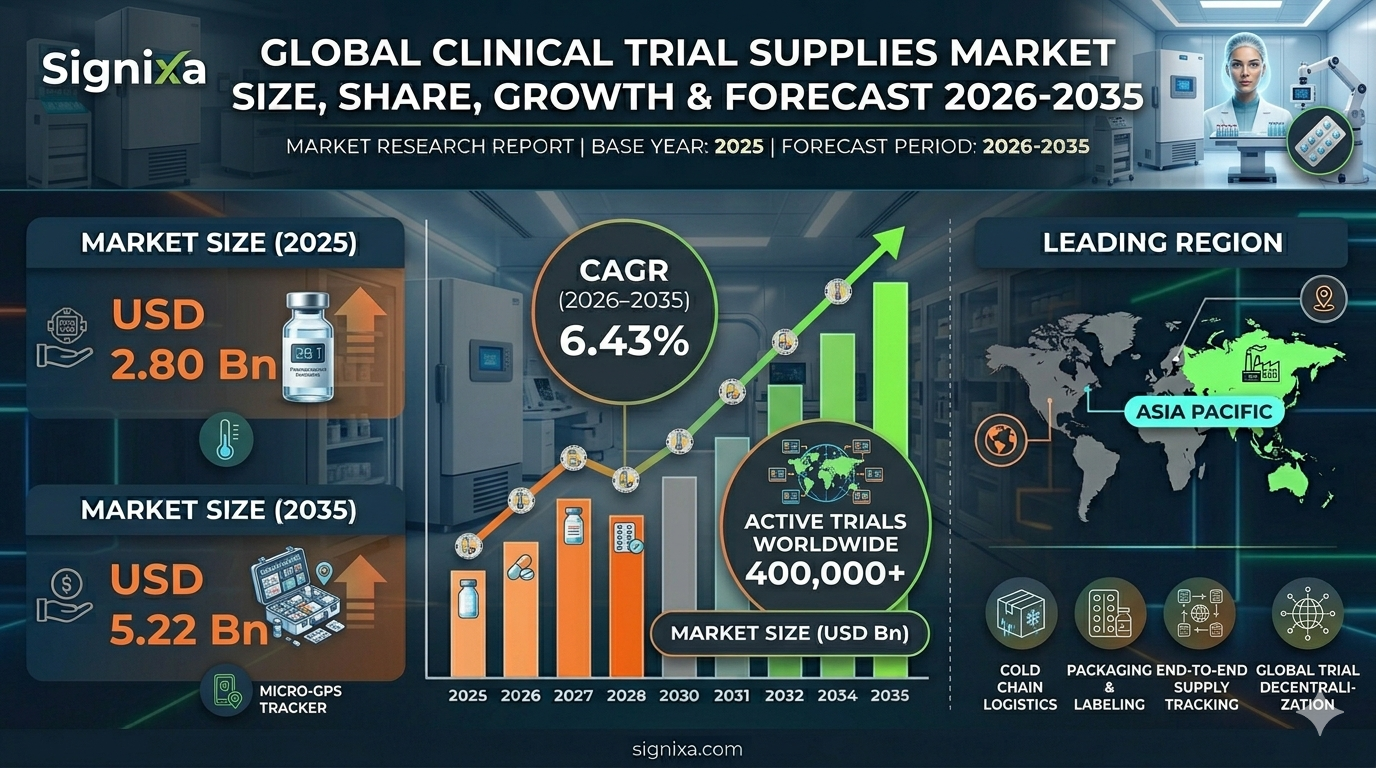
The global clinical trial supplies market was valued at approximately USD 2.80 billion in 2025 and is projected to reach around USD 5.22 billion by 2035, growing at a compound annual growth rate (CAGR) of approximately 6.43% from 2026 to 2035. This market includes a wide range of products and services essential for conducting clinical trials, such as drug substances, packaging, labeling, distribution, and logistics services. The need for faster and more efficient drug development is increasing the demand for clinical trial supplies, driven by advancements in biotechnology, personalized medicine, and regulatory frameworks.
Clinical trial supplies are necessary for conducting clinical trials for all drug development stages. They include investigational drugs, medical devices, packaging, labeling, and logistics. The market is expanding, driven by a rising number of clinical trials, advancements in biotechnology, and increasing demand for personalized medicine. As of 2025, there were over 400,000 clinical trials active worldwide and many more trials planned across all therapeutic categories. Clinical trial supplies are important to facilitate the timely and effective development of new therapies; the market is evolving to accommodate increasingly complex and personalized clinical trials.
The clinical trial supplies market is expanding due to growing investments in research and the rise of chronic diseases. Increasing demand for personalized medicine fuels this growth, with oncology, infectious diseases, and neurology as key focus areas. Efficient logistics and temperature-controlled supply chains are vital, given the globalization of trials. Advanced technologies like AI, blockchain, and IoT improve supply chain transparency and inventory management, enhancing trial efficiency. North America leads due to strong pharma and biotech sectors, while Asia Pacific grows fast, benefiting from cost advantages and supportive regulations.
Key Findings
- Phase III trials hold the largest share of the market — expected to capture over 50% market share and grow at a 10.2% CAGR from 2026-2035 — driven by large-scale trials required for drug approval.
- North America dominates the clinical trial supplies market due to its advanced clinical trial infrastructure and regulatory environment, retaining market share of over 40% through 2035.
- Oncology is the largest therapeutic segment for clinical trial supplies, accounting for over 30% of the global clinical trial supplies market due to the large volume of cancer treatment trials.
- Pharmaceutical companies are the largest end-users, anticipated to contribute over 50% of market share by 2025, performing trials across a wide range of therapeutic areas.
- The cold chain logistics segment is the fastest-growing product & services category, projected to grow at a CAGR of 11.4%, driven by globalized biologics development.
- The global clinical trial supplies market was valued at approximately USD 2.80 billion in 2025 and is projected to reach USD 5.22 billion by 2035 at a CAGR of 6.43%.
Clinical Trial Supplies Market Dynamics
Oncology & Rare Disease Drug Development — 30%+ of All Global Trials, 53 FDA Cancer Drug Approvals (2025)
A significant driver of the clinical trial supplies market is the need for new drug development in oncology, cardiology, and rare diseases. In 2025, clinical trials for the development of oncology drugs made up more than 30% of all global clinical trials. The FDA alone approved 53 new cancer drugs in 2025, which shows that innovation in this space continues to accelerate. As a result, the demand for clinical trial supplies, including investigational drugs and medical devices, has also rapidly increased to facilitate new clinical trials for these drug development efforts.
Biotechnology Innovation — Biologics, Monoclonal Antibodies & Gene Therapies Driving Specialized Supply Needs
Innovation in biotechnology is a major driver of growth in the clinical trial supplies sector. Biologic drugs, such as monoclonal antibodies and gene therapies, have unique and increased demands for highly specialized clinical trial supplies, including cold chain logistics and complex drug delivery systems. The biotechnology market is projected to grow to USD 1.2 trillion by 2035, and clinical trials will drive relevant clinically impactful biotechnology products to market. Advanced biotech products will require even more clinical trial supplies to support their complex development and approval path.
Personalized Medicine — CAGR 11.5%, Driving Demand for Individualized & Precision Clinical Trial Supply Chains
The field of personalized medicine is growing rapidly in clinical trials as a treatment modality for many diseases based on an individual's genetic makeup and environmental factors. Personalized medicine focuses on individualized and more precise treatment and requires very specific clinical trial supplies and logistics. The global personalized medicine market is projected to increase at a CAGR of 11.5%, making it one of the fastest-growing sectors of the healthcare and pharmaceutical industry. Personalized medicine is driving a need for more precise and individualized clinical trial supplies.
Emerging Market Expansion — Asia-Pacific Clinical Trial CAGR 12%, China & India Attracting Global Sponsors
Emerging markets, especially those in Asia-Pacific and Latin America, are becoming more prominent as sites for clinical trials due to the bigger pools of patients, cheaper trials, and growing healthcare systems available. The Asia-Pacific region will see the most growth in clinical trial activities, projected to have the highest CAGR of 12% through 2035. Countries such as China and India, with their large, cost-effective patient populations, are increasingly attracting clinical trial sponsors.
Clinical Trial Supplies Market Segmentation Analysis
By Clinical Phase Segment Analysis
The clinical trial supplies market is segmented by clinical phase — Phase I, Phase II, Phase III, and Phase IV. The Phase III segment holds the largest share due to the large-scale nature of trials conducted in this phase, expected to capture over 50% of the market share and grow at a 10.2% CAGR from 2026-2035.
Phase I — Safety & Dosing
Initial trials take place with a small number (20–100 participants). Their main purpose is to determine the safety of the drug, the range of doses that can be used, and what adverse reactions occur. The global early-stage trial market is projected to grow at a CAGR of 8.5% during the forecast period, driven by the increase in the number of biotech companies developing new therapies.
Phase II — Efficacy & Side Effects
Phase II trials engage a larger sample size of around 100–300 participants, with the primary objective to assess the effectiveness of the drug and its side effects. Since Phase II trials are larger and more complicated, demand for clinical trial supplies — including investigational drugs, medical devices, and patient monitoring systems — increases. CAGR expected to reach 9.3% by 2035.
Phase III — Confirmatory Large-Scale Trials
Phase III clinical trials are large trials using 300 to 3,000 individuals designed to confirm the drug's efficacy and evaluate side effects. Phase III studies require a significant amount of clinical trial supplies, including the investigational product, placebo, and any medical device. The Phase III segment is expected to capture over 50% of the market share and grow at a 10.2% CAGR from 2026-2035.
Phase IV — Post-Marketing Surveillance
Phase IV studies are conducted after a drug has reached marketing authorization. They assess long-term effects of the new drug and ensure that the drug still performs as expected in the general population. While Phase IV studies often have a smaller number of participants, they still require significant resources for monitoring and safety. Demand for clinical trial supplies in Phase IV is projected to increase at a CAGR of 7.8%.
By Product & Services Segment Analysis
Clinical Trial Materials
This includes the investigational drugs, medical devices, and diagnostic kits required to conduct the trial. These products (drugs, devices, kits, etc.) must comply with high quality standards to achieve trial integrity and protect patients' safety. The increase in number of clinical trials, especially in oncology and the biotechnology field, is driving the need for clinical trial materials. The clinical trial materials market is projected to have a CAGR of 10.1% between 2025 to 2035.
Packaging & Labelling
Packaging and labelling ensure that clinical trial materials are properly identified and handled. This includes tamper-evident packaging, packaging to allow for cold chain, and labelling that meets regulations. As clinical trials become increasingly complex, especially regarding biologics and gene therapies, the demand for specialized packaging and labelling continues to increase. This segment is expected to grow at a CAGR of 9.6%.
Cold Chain Logistics
Timely and effective distribution and logistics are essential to ensuring that clinical trial materials arrive at the site on time and in the proper condition. Cold chain logistics is especially important for biologics and gene therapies. There is an ongoing demand for efficient logistics and distribution services driven by globalized clinical trials and the increased number of biologics in clinical development. The logistics segment is projected to grow at a CAGR of 11.4%.
Other Services (Trial Management)
These solutions provide inventory management, oversight of trials, compliance with regulations, and supply chain management so that trials run as planned. In a clinical trial environment of increasing complexity, the pharmaceutical industry is increasingly turning to clinical trial management solutions to improve operational efficiency and expedite regulatory compliance. This segment is projected to achieve a CAGR of 10.3%.
By Therapeutic Area Segment Analysis
Oncology — Dominant Therapeutic Segment
Oncology clinical trials dominate the market due to the large number of new treatments being developed to treat cancer. Cancer treatment is one of the most researched areas of healthcare, which significantly influences the demand for clinical trial supplies. Oncology trials account for over 30% of the global clinical trial supplies market. The FDA approved 53 new cancer drugs in 2025 alone, underscoring the continued acceleration of innovation in this space.
Cardiovascular Diseases
Clinical trials involving drugs and devices associated with cardiology focus on treatment for heart disease, still among the leading causes of death worldwide. The high prevalence of cardiovascular disease means that cardiology trials will remain an influential market driver of clinical trial supplies demand throughout the forecast period, supporting a steady pipeline of investigational cardiovascular therapies.
Neurological Disorders
Neurology studies explore therapy for neurodegenerative disorders like Alzheimer's, Parkinson's, and multiple sclerosis. With the increasing prevalence of neurological disorders, especially across the world's older population, the desire for clinical trial supplies is continuing to grow in this area. This segment benefits from a robust pipeline of novel therapeutics targeting CNS conditions across multiple mechanisms of action.
Infectious Diseases & Others
Other therapeutic areas include infectious diseases, diabetes, and rare diseases, each contributing to the clinical trial supplies market. Due to rising global health challenges, the number of trials in these areas continues to grow, thereby driving the demand for clinical trial supplies. The COVID-19 pandemic accelerated the development of infectious disease trial infrastructure and supply chain capabilities that continue to benefit this segment.
By End-Use Segment Analysis
Pharmaceutical Companies
Pharmaceutical firms are the largest users of clinical trial supplies, performing trials for drug development in support of a wide range of therapeutic areas. The increasing pharmaceutical market, particularly due to the development of new therapies, is driving demand for clinical trial supplies. The pharmaceutical market is anticipated to contribute to over 50% of market share by 2025 and remain the dominant end-user through the forecast period.
Biopharmaceutical Companies
Biotechnology firms are primarily focused on developing biologic therapies such as monoclonal antibodies, gene therapies, and cell-based therapies — all needing specialized clinical trial supplies. With so many biologic drugs in production, biotechnology companies are seeing greater demand for clinical trial supplies. The biotechnology segment is projected to grow at a CAGR of 12.0%, the fastest among all end-user segments.
Contract Research Organizations (CROs)
CROs oversee clinical trials on behalf of pharmaceutical and biotechnology companies, and they are substantial consumers of clinical trial supplies. The expansion of pharmaceutical companies outsourcing trial management to CROs is expanding the demand for clinical trial supplies in the CRO sector. The CAGR for the CRO segment is projected to be 10.5%, reflecting the broad industry trend toward outsourced trial management.
Research Institutes & Medical Device Firms
Clinical studies for medical devices, including diagnostic tools, implants, and therapeutic devices, also require clinical trial supplies. Research institutes conducting academic and government-sponsored trials represent an additional demand pool. The increasing demand for new and advanced medical devices has contributed to the need for clinical trial supplies in this segment. The medical device sector is anticipated to grow at a CAGR of 8.0% during the forecast period.
Report Attributes & Market Scope
| Report Attribute | Details |
|---|---|
| Market Size Value in 2025 | USD 2.80 Billion |
| Market Size Value in 2035 | USD 5.22 Billion |
| CAGR (2026-2035) | 6.43% |
| Base Year Used for Estimation | 2025 |
| Historic Data | 2020–2025 |
| Forecast Period | 2026-2035 |
| Segments Covered |
Clinical Phase:
Product & Services:
Therapeutic Area:
By End-Use:
|
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Major Countries Covered | U.S., Canada, Mexico, Germany, UK, France, Italy, Spain, Russia, China, Japan, South Korea, India, Southeast Asia Countries, Brazil, Argentina, GCC Countries, Turkey, Iran, Israel, South Africa, Egypt, Nigeria, etc. |
| Key Companies Profiled | Parexel International, Catalent, Alcami Corporation, Logistical Support Services (LSS), WuXi AppTec, Lonza Group, Piramal Pharma Solutions, and PCI Pharma Services, among others. |
Clinical Trial Supplies Market Regional Analysis
The market for clinical trial supplies is growing across all regions. North America is leading the way due to its established infrastructure, while Asia-Pacific and Latin America emerging trials markets are expanding quickly, thanks to lower cost of conducting trials and large populations to participate. North America will retain market share of over 40% through 2035. Europe is projected to grow at a CAGR of 8.5%. Asia-Pacific is forecasted to grow at a CAGR of 12%. Latin America is projected to grow at a CAGR of 9.2%.
North America (Market Leader)
North America dominates the clinical trial supplies market, driven by its well-established pharmaceutical and biotechnology industries. The region benefits from substantial investments in research and development, a large number of ongoing clinical trials, and advanced healthcare infrastructure. The U.S. leads with strong regulatory support from agencies like the FDA, which facilitates streamlined approval processes and ensures high standards in trial execution. The presence of numerous CROs and specialized logistics providers enhances the efficient management of clinical trial supplies, including temperature-controlled shipments. Technological adoption, such as AI-powered inventory tracking and blockchain for supply chain transparency, is prominent, improving trial accuracy and compliance.
Europe
Europe holds a significant position in the clinical trial supplies market, supported by its robust pharmaceutical industry and strict regulatory environment. The presence of the European Medicines Agency (EMA) ensures rigorous standards for clinical trial conduct and supply management. Countries such as Germany, the UK, France, and Italy are key contributors due to their advanced healthcare systems and strong clinical research ecosystems. Increasing focus on personalized medicine and rare diseases drives demand for innovative clinical trial supplies, especially in oncology and immunology sectors. Regulatory harmonization efforts across the EU aim to simplify trial approvals and supply processes, enhancing market growth.
Asia-Pacific (Fastest Growing)
Asia-Pacific appears to be the fastest-growing market for clinical trials, with countries like China and India being hotspots with large patient populations and low costs of conducting trials. The Asia-Pacific market for clinical trial supplies is forecasted to grow at a CAGR of 12%. Countries such as China, Japan, South Korea, and India are investing heavily in healthcare infrastructure and clinical research capabilities, making the region increasingly attractive to global trial sponsors seeking cost-effective, large-scale patient recruitment and regulatory-supportive environments.
Latin America
The Latin America market is emerging as an attractive market for conducting clinical trials, mainly due to cost opportunities and growing healthcare initiatives. The Latin America market for clinical trial supplies is projected to grow at a CAGR of 9.2%. Brazil and Mexico are the primary contributors, each benefiting from government-backed healthcare expansion programs, growing CRO presence, and an increasing willingness among patients to participate in clinical research. The region's cost-competitive environment compared to North America and Europe makes it an attractive destination for phase II and III trials.
Middle East & Africa
The Middle East and Africa represent an emerging market for clinical trial supplies, benefiting from increasing investment in healthcare infrastructure and a growing recognition of the value of clinical research. GCC countries, particularly Saudi Arabia and the UAE, are investing in world-class medical facilities and clinical research hubs. African nations are expanding their healthcare systems and regulatory frameworks to attract more clinical trial activity, particularly in infectious disease and tropical medicine research, creating gradual but sustained demand for clinical trial supply chain services.
Recent Developments in the Clinical Trial Supplies Market
-
April 2025
Thermo Fisher Scientific reported stronger-than-expected earnings for Q1 2025, driven by steady demand for its tools and services used in clinical research and therapy development. The company earned $5.15 per share on an adjusted basis, surpassing analysts' projections. This performance underscores the sustained demand for clinical trial infrastructure and supply services as the global drug development pipeline remains robust.
-
February 2025
FedEx Corp. unveiled its Life Science Centre in Mumbai, setting a benchmark in the clinical trial supply chain in India and globally. This facility aims to enhance the logistics and distribution capabilities for clinical trial supplies, ensuring efficient and compliant delivery of investigational products. The facility directly supports India's growing role as a key clinical trial destination, providing world-class cold chain logistics and specialized handling for temperature-sensitive biologics and investigational therapies.
Clinical Trial Supplies Market: Competitive Landscape
The clinical trial supplies market is an extremely competitive market, with many companies involved in providing materials, logistics, packaging, and management services. Some of the major companies include Parexel International Corporation, Catalent, Inc., Lonza Group, WuXi AppTec, Medpace, and PAREXEL International. These companies lead the market by offering a wide range of services and products that are customized for the needs of specific clinical trials. The market relies on strategic partnerships, mergers, and acquisitions to add value through extending capabilities in new markets and through new technologies that can help in the adoption of advanced clinical trial management design using AI and blockchain. Additionally, the continued adoption of personalized medicine and biologic treatments keeps companies innovating in providing specialized clinical trial supplies to meet the new demands generated from these therapies.
Key Companies Profiled
Global Clinical Trial Supplies Market Segmentation Summary
By Clinical Phase
By Product & Services
By Therapeutic Area
By End-Use
By Region
Frequently Asked Questions (FAQs) — Clinical Trial Supplies Market
What are clinical trial supplies?
Clinical trial supplies are the materials required for the efficient conduct of clinical trials, including investigational drugs, devices, packaging, labelling, storage solutions, and logistical services. These supplies ensure that clinical trials run smoothly, meeting regulatory and safety requirements while maintaining the integrity of the trial process.
Which key factors will influence the clinical trial supplies market growth over 2026-2035?
The global clinical trial supplies market is expected to grow due to an increasing number of clinical trials, outsourcing to Contract Research Organizations (CROs), advancements in biologics and biotechnology, and the growing demand for specialized logistics services to handle temperature-sensitive materials, especially for biologic drugs.
What will be the value of the clinical trial supplies market during 2026-2035?
According to market research, the global clinical trial supplies market was valued at around USD 2.80 billion in 2025 and is predicted to grow to approximately USD 5.22 billion by 2035, reflecting a CAGR of 6.43% during the forecast period.
What will be the CAGR value of the clinical trial supplies market during 2026-2035?
The CAGR value of the clinical trial supplies market is expected to be around 6.43% during 2026-2035, driven by factors such as the increasing number of clinical trials, rising complexity of drug development, and growing outsourcing to specialized service providers.
Which region will contribute notably towards the clinical trial supplies market value?
The North American region is expected to contribute significantly to the clinical trial supplies market, retaining over 40% market share through 2035, with the United States leading the way due to its well-established healthcare infrastructure, a large number of clinical trials, and favourable regulatory environment. Asia-Pacific is the fastest-growing region at a CAGR of 12%.
Which are the major players leveraging the clinical trial supplies market growth?
The clinical trial supplies market is led by key players such as Parexel International, Catalent, Alcami Corporation, Logistical Support Services (LSS), WuXi AppTec, Lonza Group, Piramal Pharma Solutions, and PCI Pharma Services. These companies play a significant role in providing clinical trial materials, packaging, logistics, and trial management services.
What can be expected from the global clinical trial supplies market report?
The report explores crucial aspects of the clinical trial supplies market, including a detailed discussion of existing growth factors and restraints, analysis of future growth opportunities and challenges, segmentation by clinical phase, product & services, therapeutic area, and end-use, and regional and competitive landscape insights covering the forecast period 2025 to 2035.

